Pyrogens are fever-inducing substances derived from pathogens or even abiotic materials. The presence of pyrogenic contaminants in injectables or on medical devices can lead to severe reactions of the immune system, which can cause the death of patients, particularly in seriously ill patients.
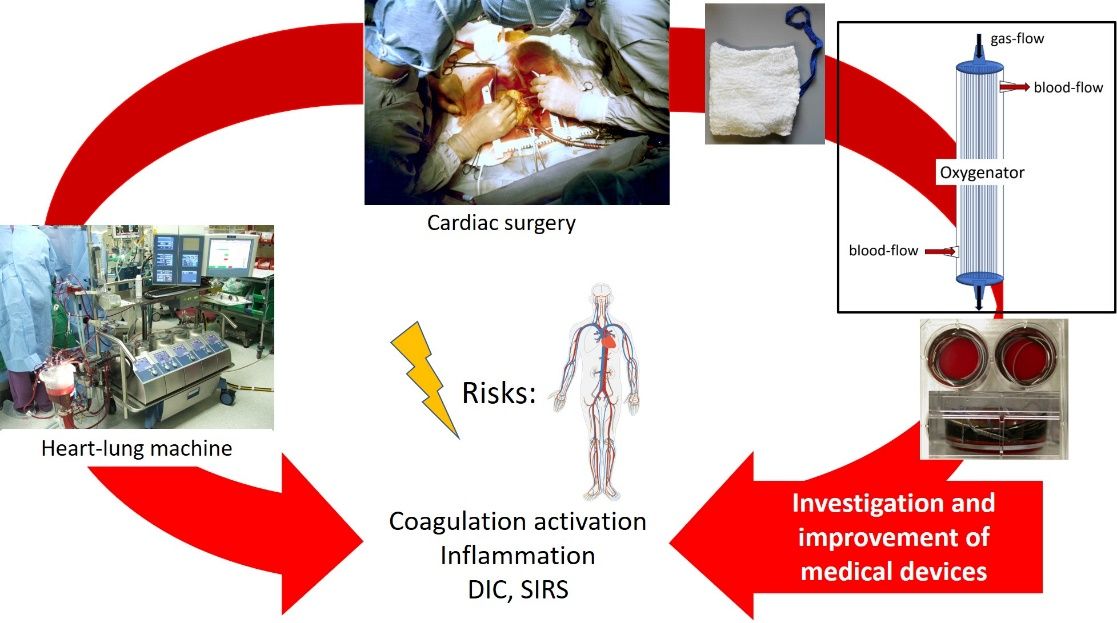
The research group focusses on the one hand on the testing of injectables and medical devices for their pyrogenic potential in vitro to prevent harmful reactions in vivo (see Figure). On the other hand, we also investigate the performance of medical devices in terms of their bio- and haemocompatibility. Our research focusses in particular on the early activation of the plasma contact phase system and the adsorption of plasma proteins. We firstly aim to understand the mechanism behind the unwanted activation and then establish methods to prevent or at least reduce the reaction leading to the activation of plasma proteins and cells, which ultimately cause the failure of the device.